Research groups
Colleges
Manuela Zaccolo
MD, PhD, FRSB
Deputy Head of Department
- Director, Burdon Sanderson Cardiac Sciences Centre
- Professor of Cell Biology
- Director, Centre for Integrative Physiology
Manuela Zaccolo graduated in medicine at the University of Torino, Italy, and subsequently went on to pursue a career in science by spending four years as a post-doctoral researcher at the LMB, MRC, Cambridge, UK, working on protein engineering and in vitro molecular evolution. She then moved back to Italy, at the University of Padova, to work on the generation of fluorescent sensors for real time imaging of intracellular second messengers in living cells. In Padova she established her independent research group in 2001 at the Venetian Institute of Molecular Medicine with a focus on intracellular signalling. In 2007 she moved to the University of Glasgow, where she initially held a position as a Reader and subsequently as Professor of Cell Biology. In 2012 she joined the Department of Physiology, Anatomy and Genetics at Oxford University. She is also a Fellow in Pre-Clinical Medicine at Balliol College.
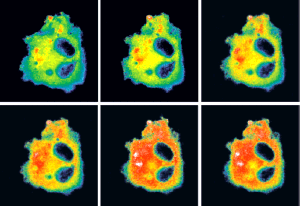
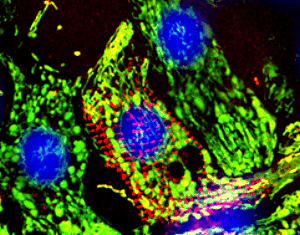
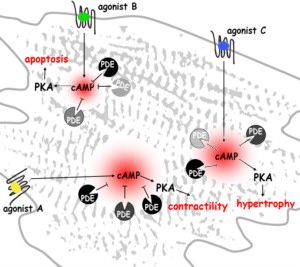
Manuela Zaccolo's research focuses on how cells sense external stimuli and how these are processed to produce a functional outcome. She is interested in the architectural and regulatory principles by which intracellular signalling networks achieve the plasticity and context-sensitivity necessary for a cell to function. In particular, her work has focused on cyclic nucleotide signalling in the heart and in other cell systems and on the role of local regulation and compartmentalisation of signal transduction in determining the specificity of cellular responses. Her ultimate goal is to understand how alteration of signalling networks leads to human disease and to apply this knowledge to the development of novel therapeutic strategies.
Further information can be found at Zaccolo Research Group.
Key publications
Integrated Proteomics Unveils Nuclear PDE3A2 as a Regulator of Cardiac Myocyte Hypertrophy.
Journal article
Subramaniam G. et al, (2023), Circ Res, 132, 828 - 848
FRET biosensor uncovers cAMP nano-domains at β-adrenergic targets that dictate precise tuning of cardiac contractility.
Journal article
Surdo NC. et al, (2017), Nat Commun, 8
Cardiac Hypertrophy Is Inhibited by a Local Pool of cAMP Regulated by Phosphodiesterase 2.
Journal article
Zoccarato A. et al, (2015), Circ Res, 117, 707 - 719
Discrete microdomains with high concentration of cAMP in stimulated rat neonatal cardiac myocytes.
Journal article
Zaccolo M. and Pozzan T., (2002), Science, 295, 1711 - 1715
A genetically encoded, fluorescent indicator for cyclic AMP in living cells.
Journal article
Zaccolo M. et al, (2000), Nat Cell Biol, 2, 25 - 29
Recent publications
Rational discovery of therapeutic PAK1 allosteric activators.
Journal article
He Y. et al, (2026), Cell
Electromechanical modelling and simulation of human-induced pluripotent stem cell-derived cardiomyocytes predict drug-induced contractility effects.
Journal article
Folkmanaite M. et al, (2026), J Physiol
Compartmentalisation in cAMP signalling: A phase separation perspective.
Journal article
Folkmanaite M. and Zaccolo M., (2025), Br J Pharmacol
GPCR signaling via cAMP nanodomains.
Journal article
Yadav R. and Zaccolo M., (2025), Biochem J, 482, 519 - 533