Research groups
Websites
-
Kavli Institute for Nanoscience Discovery webpage
Kavli Profile
-
PubMed
Publications
-
ORCID profile
Publication list
-
OPDC
Oxford Parkinson's Disease Centre
Brent Ryan
Group Leader, Departmental Research Lecturer
- Research Scientist
Research Summary
Following his undergraduate and doctoral training at the University of Exeter, Dr. Brent Ryan joined the University of Oxford in 2010 as a member of the Oxford Parkinson’s Disease Centre. His lab's work focuses on understanding the molecular pathways that make certain neurons vulnerable in Parkinson’s disease, using patient-derived induced pluripotent stem cell (iPSC) models.
The Ryan Laboratory uses high-throughput biology and advanced proteomics to investigate how mitochondrial and lysosomal dysfunction contribute to neurodegeneration. A core part of the lab’s mission is to design and develop innovative assays that measure key aspects of neuronal health, organelle function, and cellular stress. These tools enable the team to study how both genetic risk factors and environmental stressors shape disease processes in human neurons.
The lab has established powerful high-throughput and high-content phenotypic screening platforms that combine CRISPR interference (CRISPRi) and diverse compound libraries with mature patient-derived iPSC neurons, primary rodent neurons, and engineered cell lines. By integrating automated imaging, functional measurements, and large-scale data analysis, the Ryan Lab identifies molecular pathways that drive neurodegeneration and reveals new opportunities for therapeutic intervention.
Working closely with academic and industry partners, the laboratory aims to translate fundamental discoveries into strategies that support the development of disease-modifying treatments for Parkinson’s disease and related neurological disorders.
Biography
I completed a PhD at University of Exeter (Peninsula Medical School) in which I studied the effects of oxidative post-translational modifications on the breakdown of immune tolerance in the autoimmune disease systemic lupus erythematosus. I joined the Wade-Martins lab in 2009 and initially worked on the effects of oxidative stress and mitochondrial dysfunction on cellular biochemistry in toxin models of PD and how α-synuclein levels impact these. In 2015 I became an OPDC Career Development Fellow and am focussing on developing high-throughput screens for Parkinson's disease. I was awarded a MRC New Investigator Research Grant in 2023.
Recent publications
Phosphorylated ubiquitin is a secondary messenger and an epigenetic mark mediating mitochondria to nucleus signaling.
Preprint
Mercer TJ. et al, (2026)
CHCHD2 links mitochondrial dysfunction and α-synuclein misfolding in Parkinson's disease.
Journal article
Narendra D. and Ryan BJ., (2026), Trends Neurosci, 49, 157 - 158
M1-linked ubiquitination by LUBAC regulates AMPK signalling and the response to energetic stress.
Journal article
Elbæk CR. et al, (2026), Cell Death Differ
Heritable maintenance of chromatin modifications confers transcriptional memory of interferon-γ signaling.
Journal article
Mikulski P. et al, (2025), Nat Struct Mol Biol, 32, 1255 - 1267
Formation of seeding-competent α-synuclein aggregates in parkin-deficient iPSC-derived human neurons.
Journal article
Schmidt SI. et al, (2025), NPJ Parkinsons Dis, 11
TFEB and TFE3 have cell-type specific expression in the brain and divergent roles in neurons
Preprint
McGuinness W. et al, (2025)
iPSC-derived dopaminergic culture
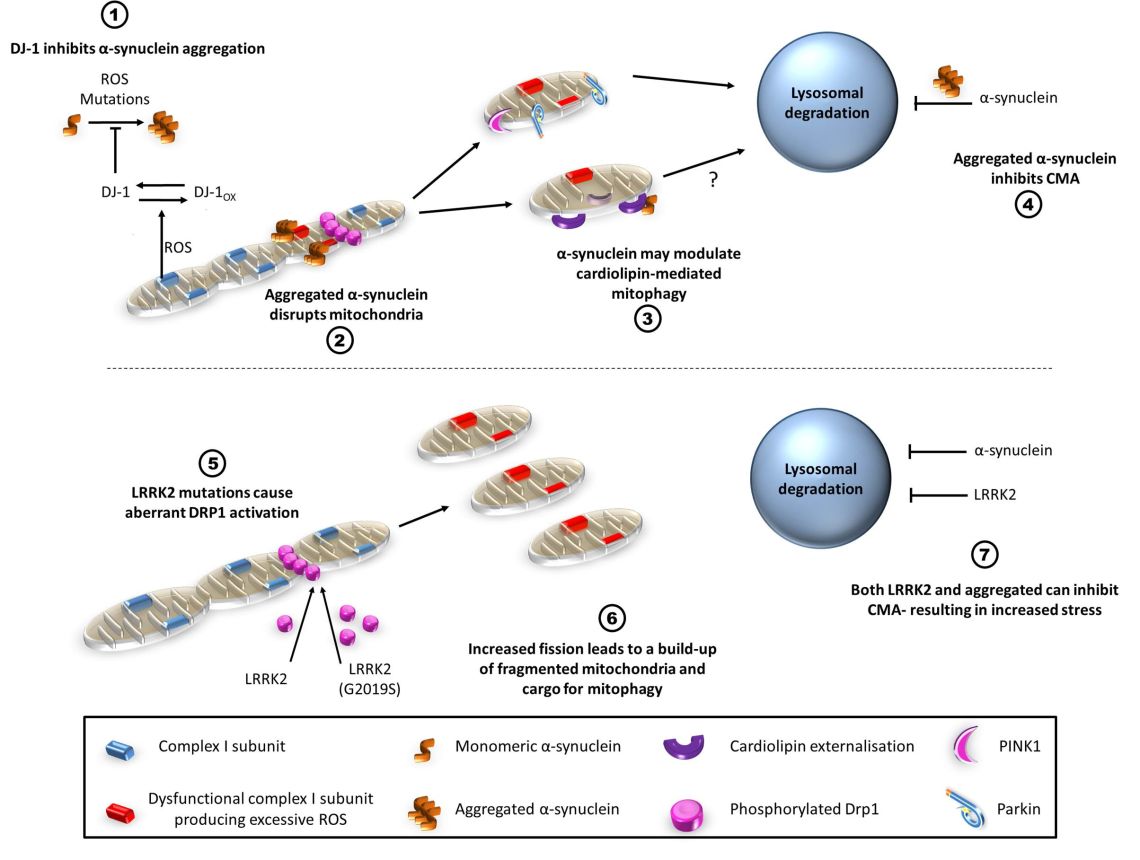
Roles of alpha-synuclein and LRRK2 in mitophagy